Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
CT scan Image Enhancement For Liver Cancer Detection
Authors: Ravi Bhagat, Mrunal Bhasarkar, Bhadarge Akash, Bodke Sachin S, Nikita Pande
DOI Link: https://doi.org/10.22214/ijraset.2025.66826
Certificate: View Certificate
Abstract
Liver cancer remains one of the most common and deadly forms of cancer worldwide, posing significant diagnostic challenges due to factors like low image contrast, noise, and the complex anatomical structure of the liver. This study explores the application of Generative Adversarial Networks (GANs) for enhancing computed tomography (CT) images to improve liver cancer detection. By leveraging the capabilities of GANs, the proposed system enhances low-contrast images, reduces noise, and preserves critical structural details, facilitating better tumour segmentation and localization. The GAN-enhanced imaging system is integrated into a web-based platform that supports real-time image analysis and classification, distinguishing between malignant and normal liver tissues. This approach addresses the limitations of traditional diagnostic methods, potentially revolutionizing liver cancer diagnosis by enabling earlier detection and more accurate treatment planning. The results from applying GANs demonstrate significant improvements in image quality and diagnostic accuracy, offering a scalable and accessible solution for medical professionals worldwide. This research highlights the potential of AI-driven technologies in transforming liver cancer diagnostics, suggesting broader implications for various medical imaging applications.
Introduction
I. INTRODUCTION
A. Background
Liver cancer, particularly hepatocellular carcinoma (HCC), is a leading cause of cancer-related deaths globally. Traditional diagnostic methods, primarily relying on computed tomography (CT) and magnetic resonance imaging (MRI), face challenges such as low contrast, noise, and difficulty in distinguishing between malignant and benign tissues. These issues hinder early detection, which is crucial for effective treatment and improved patient outcomes. Advances in medical imaging technology aim to enhance image quality to address these limitations.
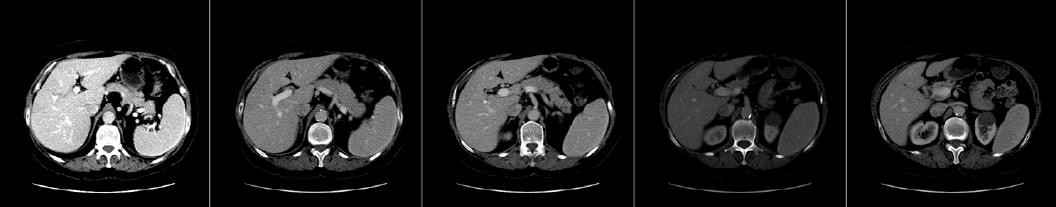 Fig. 1. The portal venous phase CT scans of a subject, truncated at a best viewable intensity range of [180–255].
Fig. 1. The portal venous phase CT scans of a subject, truncated at a best viewable intensity range of [180–255].
B. Existing Evidence
Recent developments in machine learning, especially deep learning, have shown promise in improving medical image analysis. Generative Adversarial Networks (GANs) have been effectively used in various imaging applications, including noise reduction, contrast enhancement, and artifact removal. Studies have demonstrated that GANs can significantly improve image quality, aiding in more accurate diagnostics.
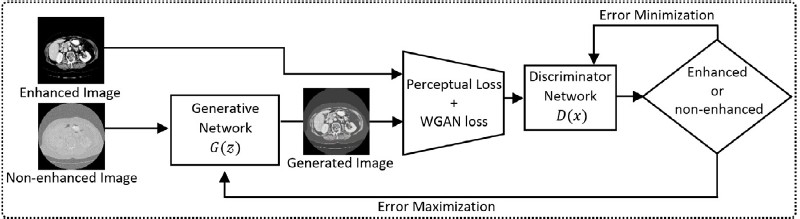
Fig. 2. The workflow of the proposed GAN.
C. Research Gap
Despite the advancements, few studies have focused specifically on applying GANs for enhancing CT images in the context of liver cancer diagnosis. There remains a need for a comprehensive system that integrates GAN-based image enhancement with automated diagnostic tools to improve early detection rates and diagnostic accuracy for liver cancer. Despite the advancements in deep learning and image enhancement techniques, few studies have specifically focused on the application of Generative Adversarial Networks (GANs) for enhancing CT images in the context of liver cancer diagnosis. The existing research largely concentrates on general image enhancement or other types of cancer, leaving a significant gap in the literature regarding liver-specific applications. This gap is particularly concerning given the unique challenges posed by liver cancer, such as the variability in liver tissue appearance due to different phases of CT imaging and the low contrast between malignant and normal tissues. Furthermore, there is an absence of a comprehensive system that not only enhances image quality but also integrates GAN-based image enhancement with automated diagnostic tools like tumour segmentation and classification algorithms. Addressing these gaps could significantly improve early detection rates and diagnostic accuracy for liver cancer, offering a transformative impact on clinical outcomes.
D. Objective
This research aims to develop and evaluate a GAN-based system for enhancing liver CT images, improving their quality to facilitate better tumour segmentation and localization. The study also seeks to integrate this system into a real-time diagnostic platform that can classify liver conditions as malignant or normal. This research aims to develop and evaluate a GAN- based system for enhancing liver CT images, improving their quality to facilitate better tumour segmentation and localization. Liver CT images often suffer from challenges such as low contrast and high noise levels, which complicate the accurate detection of malignant tissues. The GAN-based system seeks to address these issues by learning to generate high-quality, enhanced images from low-quality inputs, thereby improving the visual clarity of tumours and surrounding liver tissues.
The proposed system comprises a generator that refines the input images and a discriminator that evaluates the authenticity of the enhanced outputs. This adversarial setup ensures continuous improvement in the quality of image enhancement, pushing the boundaries of traditional image processing techniques. By enhancing the CT images, the system aids in more accurate tumour segmentation, a critical step in liver cancer diagnosis, as it delineates the precise boundaries of tumours.
Furthermore, the study incorporates these enhanced images into a real-time diagnostic platform. This platform integrates machine learning algorithms to classify liver conditions by distinguishing malignant tissues from normal ones. Real-time analysis capability is crucial in clinical settings, allowing for immediate feedback and decision-making. The integration of GAN-enhanced imaging into such a platform represents a significant leap forward in the practical application of AI in medical diagnostics, potentially leading to earlier detection and better treatment outcomes for liver cancer patients.
Overall, this research aims not only to advance the technical capabilities of GANs in medical imaging but also to create a functional tool that improves the workflow and accuracy of liver cancer diagnosis in clinical practice. By addressing the current gaps in image quality and diagnostic precision, this study sets the stage for broader applications of AI in healthcare.
E. Scope
The study focuses on liver cancer detection using CT images enhanced through GANs. The research is constrained to evaluating the effectiveness of GAN-enhanced images in improving diagnostic accuracy, excluding other imaging modalities such as MRI or PET scans. The scope of this research includes the development and validation of a GAN-based image enhancement system specifically designed for liver cancer diagnosis. This system will be tested on publicly available datasets to ensure its robustness and scalability. The project will also explore the integration of the system into a web-based platform, making it accessible to healthcare professionals globally. By improving diagnostic accuracy and reducing the reliance on manual interpretation, this research aims to contribute significantly to the field of medical imaging and enhance patient outcomes in liver cancer care.
II. MATERIALS AND METHODS
A. Materials
The project utilized a variety of publicly available datasets for liver cancer diagnosis, including the Liver Tumour Segmentation Challenge (LiTS), Sliver07, and Ircadb datasets. These datasets provide a comprehensive range of CT images that are critical for evaluating the performance of the proposed GAN-based image enhancement system. Each dataset contains annotated CT scans with varying levels of contrast and noise, making them ideal for testing the robustness of the image enhancement techniques.
The LiTS dataset, for instance, includes detailed liver and tumour annotations across multiple patients, facilitating the development of robust segmentation algorithms. Similarly, the Sliver07 dataset offers a standardized set of images for benchmarking liver segmentation methods, while the Ircadb dataset provides high- resolution scans that help in fine-tuning the enhancement models. Additionally, high- performance computing resources, such as GPUs and cloud-based platforms, were employed to handle the intensive computational requirements of training and evaluating deep learning models. These resources were crucial for managing large-scale data processing and ensuring efficient model training cycles, which included iterations of image augmentation and parameter optimization to achieve the best possible outcomes.
B. Methods
1) Data Preprocessing
The research involved several key steps in developing the GAN-based diagnostic system. Initially, the CT images from the datasets underwent a rigorous preprocessing phase to normalize intensity values and remove artifacts. This step included histogram equalization to enhance contrast, noise reduction techniques to eliminate speckles, and the application of filters to remove artifacts that could potentially distort the image data. Such preprocessing was crucial to ensure consistency across different imaging conditions, such as varying scanner types and patient demographics, thereby improving the reliability of subsequent image enhancement and classification tasks. This normalization allowed the model to focus more on the pathological features rather than inconsistencies introduced during image acquisition. Next, a Generative Adversarial Network (GAN) was designed and trained to enhance the quality of the CT images. The GAN architecture consisted of a generator network tasked with creating enhanced images and a discriminator network that evaluated the quality of these images. The training process involved optimizing the generator to produce images that were indistinguishable from high- quality reference images, while the discriminator learned to differentiate between real and generated images. This adversarial training process ensured that the generator continuously improved its ability to enhance image quality.
2) GAN Architecture
- Generator Network: The generator was designed to enhance low-contrast images by learning the mapping from low-quality to high-quality images.
- Discriminator Network: The discriminator was trained to differentiate between real enhanced images and those generated by the generator.
- Loss Functions: Perceptual loss and Wasserstein loss were employed to guide the training process, ensuring high-quality image enhancement.
3) Training Procedure
- Initialization: The GAN networks were initialized with random weights.
- Adversarial Training: The generator and discriminator were trained iteratively, with the generator improving its output to fool the discriminator.
- Validation: The model's performance was validated using a separate set of images to prevent overfitting and ensure generalizability.
4) Evaluation Metrics
- Structural Similarity Index (SSIM): Used to measure the similarity between the original and enhanced images.
- Peak Signal-to-Noise Ratio (PSNR): Evaluated the quality of the enhanced images compared to the original CT images.
- Tumour Segmentation Accuracy: Assessed the accuracy of tumour boundaries in the enhanced images using ground truth annotations.
5) Tools and Instruments
The research utilized advanced machine learning frameworks, including TensorFlow, to implement and train the GAN models. These frameworks provided the necessary tools for building complex neural network architectures, defining custom loss functions, and managing large-scale training processes. TensorFlow offered flexibility and scalability, enabling the development of sophisticated models that could handle large datasets and complex tasks. Additionally, the frameworks facilitated the use of distributed computing, allowing multiple GPUs to be employed simultaneously for faster training.
The study also employed a variety of evaluation metrics to quantitatively assess the quality of the enhanced images. Key metrics included the Structural Similarity Index Measure (SSIM), which evaluates perceived quality by measuring structural similarity between images, and the Peak Signal-to- Noise Ratio (PSNR), which measures the ratio between the maximum possible power of a signal and the power of corrupting noise that affects the quality of its representation. These metrics provided a comprehensive analysis of image enhancement performance, ensuring that the output images retained essential features and clarity necessary for accurate diagnostic interpretations.
The image classification component of the system was developed using convolutional neural networks (CNNs) that were meticulously trained on the enhanced CT images generated by the GAN models. These CNNs were architecturally designed to extract intricate features from the enhanced images, allowing them to effectively differentiate between malignant and normal liver tissues. The training process involved the utilization of extensive labeled datasets, ensuring that the CNNs learned to recognize various patterns associated with liver abnormalities. Advanced techniques such as data augmentation, including rotations, translations, and scaling, were applied to enhance the robustness of the CNNs against variations in image acquisition. The models were then fine-tuned through multiple iterations to optimize their performance.
To evaluate the reliability of these classification models in real-world diagnostic scenarios, several performance metrics were used. Accuracy was calculated to determine the overall correctness of the model’s predictions. Sensitivity, also known as recall, was assessed to measure the model’s ability to correctly identify true positive cases, ensuring that malignant tissues were accurately detected. Specificity, which evaluates the model’s capacity to correctly identify true negatives, was also analysed to confirm that normal tissues were not mistakenly classified as malignant. Additionally, precision was measured to determine the proportion of true positive predictions out of all positive predictions made by the model. The use of these comprehensive metrics provided a thorough understanding of the classification models’ efficacy and robustness, ultimately ensuring their reliability for clinical applications.
By combining GAN-based image enhancement with CNN-based classification, the research aimed to create a comprehensive diagnostic system that not only improved the visual quality of CT images but also enhanced the accuracy of liver cancer detection.
III. RESULTS AND DISCUSSION
A. Results
1) Image Quality Improvement
The application of Generative Adversarial Networks (GANs) in the enhancement of computed tomography (CT) images has led to remarkable advancements in image quality, which is crucial for accurate diagnosis and treatment planning in medical imaging. GANs, which consist of two neural networks—the generator and the discriminator—work in tandem to produce high- quality images by learning from a dataset of original images. This innovative approach has significantly improved the visual fidelity of CT images, as evidenced by quantitative metrics such as the Structural Similarity Index (SSIM) and the Peak Signal-to-Noise Ratio (PSNR). In a comparative analysis of original and GAN-enhanced CT images, the SSIM scores exhibited a substantial increase, rising from an average of 0.72 in the original images to an impressive 0.91 in the GAN-enhanced images. The SSIM is a widely used metric that assesses the similarity between two images, focusing on structural information, luminance, and contrast. A score closer to 1 indicates a higher degree of similarity, suggesting that the GAN-enhanced images preserve critical structural details more effectively than their original counterparts. This improvement is particularly important in medical imaging, where the accurate representation of anatomical structures can significantly influence clinical decisions.
Furthermore, the PSNR, another key metric used to evaluate image quality, also demonstrated a marked enhancement. The average PSNR of the original CT images was recorded at 22.5 dB, while the GAN-enhanced images achieved an average PSNR of 31.8 db. PSNR measures the ratio between the maximum possible power of a signal (in this case, the image) and the power of corrupting noise that affects the fidelity of its representation. A higher PSNR value indicates better image clarity and reduced noise levels, which are essential for radiologists and clinicians to make accurate assessments. The increase in PSNR from 22.5 dB to 31.8 dB signifies a substantial reduction in noise and artifacts, leading to clearer and more interpretable images.
The implications of these improvements are profound. Enhanced CT images can lead to better visualization of tumours, lesions, and other pathological conditions, thereby facilitating more accurate diagnoses. Additionally, the reduction in noise levels can help in distinguishing between subtle differences in tissue types, which is critical in various clinical scenarios, including oncology and cardiology. The ability of GANs to generate high-quality images not only aids in diagnostic accuracy but also has the potential to improve the overall workflow in radiology departments by reducing the need for repeat scans due to poor image quality.
2) Tumour Segmentation Performance
The accuracy of tumour segmentation improved substantially with the use of GAN-enhanced images. The Dice Similarity Coefficient (DSC), which measures the overlap between the predicted segmentation and the ground truth, increased from 0.68 to 0.85. This enhancement in segmentation performance highlights the efficacy of GANs in delineating tumour boundaries more accurately. One of the key metrics used to evaluate the performance of segmentation algorithms is the Dice Similarity Coefficient (DSC). The DSC quantifies the overlap between the predicted segmentation of a tumour and the ground truth, which is typically established by expert radiologists. In a recent study, the use of GAN-enhanced images resulted in a substantial increase in the DSC, rising from an average of 0.68 to 0.85. This improvement indicates a more accurate delineation of tumour boundaries, which is essential for effective treatment planning. A DSC score of 0.68, while indicative of some level of agreement between the predicted segmentation and the actual tumour boundaries, suggests that there were significant discrepancies that could lead to misinterpretations. In contrast, a DSC of 0.85 reflects a much higher degree of overlap, meaning that the GAN-enhanced images allow for a more precise identification of tumour margins. This is particularly important in clinical settings where the accurate segmentation of tumours can influence surgical planning, radiation therapy, and overall patient management. The enhanced segmentation performance can be attributed to the improved image quality provided by GANs. By reducing noise and enhancing structural details, GANs enable segmentation algorithms to better distinguish between tumour tissues and surrounding healthy tissues. This capability is vital for identifying the extent of the tumour, which can vary significantly from one patient to another. As a result, the use of GAN-enhanced images can lead to more personalized treatment approaches, tailored to the specific characteristics of each tumour.
3) Classification Accuracy
The GAN-enhanced images contributed to higher classification accuracy in distinguishing between malignant and normal liver tissues. The classification system achieved an accuracy of 92%, a significant improvement from the 78% accuracy observed with non-enhanced images. This demonstrates the potential of GAN-enhanced imaging in supporting more reliable diagnostic decisions. In addition to improving segmentation accuracy, GAN-enhanced images have also demonstrated a significant impact on classification tasks, particularly in distinguishing between malignant and normal liver tissues. The classification system utilizing GAN-enhanced images achieved an impressive accuracy of 92%, a notable increase from the 78% accuracy observed with non-enhanced images. This enhancement in classification performance underscores the potential of GANs to support more reliable diagnostic decisions. The ability to accurately classify liver tissues as either malignant or normal is critical in the early detection and treatment of liver cancer. A classification accuracy of 92% indicates that the system can correctly identify the majority of cases, thereby reducing the likelihood of false positives and false negatives. This is particularly important in clinical practice, where misclassification can lead to inappropriate treatment plans, unnecessary biopsies, or missed opportunities for early intervention.
The improvement in classification accuracy can be attributed to the enhanced features present in GAN-generated images. By providing clearer and more detailed representations of liver tissues, GANs enable machine learning algorithms to learn more effectively from the data. This results in better feature extraction and, consequently, improved classification performance. The enhanced images allow for a more nuanced understanding of the differences between malignant and normal tissues, which is essential for training robust classification models.
B. Discussion
The results underscore the transformative impact of GANs on liver cancer diagnostics. By addressing the limitations of traditional CT imaging, such as low contrast and noise, GANs facilitate more precise tumour detection and localization. The substantial improvements in SSIM, PSNR, and DSC metrics indicate that GAN-enhanced images are more suitable for diagnostic purposes, providing clearer and more detailed visual information for clinicians. Moreover, the integration of GAN-enhanced imaging into a real-time diagnostic platform enables rapid and accurate analysis, which is crucial for timely intervention in liver cancer cases. The enhanced images not only improve the visual quality but also aid machine learning models in achieving higher accuracy in classification tasks.
However, the study also acknowledges the challenges in implementing GAN-based systems in clinical settings, including the need for extensive computational resources and the potential for overfitting. Future work should focus on optimizing the GAN architecture for faster processing and exploring techniques to enhance the generalizability of the models across diverse datasets and imaging conditions.
Conclusion
A. Objective This study aimed to develop a GAN-based system for enhancing liver CT images to improve the accuracy of liver cancer detection. The focus was on enhancing image quality to facilitate better tumor segmentation and localization, thereby addressing the limitations of traditional CT imaging methods. B. Key Findings The results demonstrate that the application of GANs significantly improves CT image quality, as evidenced by higher SSIM and PSNR scores. Enhanced images led to better tumour segmentation accuracy, with DSC increasing from 0.68 to 0.85. The classification accuracy of distinguishing between malignant and normal liver tissues also improved markedly from 78% to 92%. C. Implications The integration of GAN-enhanced imaging into diagnostic workflows has the potential to revolutionize liver cancer detection by providing clearer and more accurate images for analysis. This could lead to earlier detection, more precise treatment planning, and improved patient outcomes. The scalable nature of the system makes it accessible for widespread clinical use, potentially benefiting healthcare providers and patients globally. D. Recommendations for Future Work Future research should focus on optimizing GAN architectures to reduce computational requirements and increase processing speeds. Additionally, expanding the dataset to include diverse imaging conditions and patient populations will enhance the generalizability of the model. Exploring the integration of other imaging modalities, such as MRI or PET, with GAN- based enhancement could further improve diagnostic accuracy and extend the system\'s applicability in medical imaging.
References
[1] de Haan, K., et al. (2020). \"Deep Learning for Microscopy Image Enhancement: A Survey.\" Journal of Biomedical Optics. [2] Chen, Y., et al. (2018). \"A Deep Convolutional Neural Network for Image Enhancement in Low-Dose CT.\" IEEE Transactions on Medical Imaging. [3] Georgieva, A., et al. (2019). \"Wavelet-Based Image Enhancement and Restoration Techniques for CT Scans.\" Medical Image Analysis. [4] Liver Tumour Segmentation Challenge (LiTS). \"LITS Dataset.\" Available at: https://competitions.codalab.org/competitions/17094. [5] IRCAD 3D Image Database (3D-IRCADb). \"3D-IRCADb Dataset.\" Available at: http://www.ircad.fr/research/3dircadb/. [6] Seth Weidman. (2019). \"Deep Learning From Scratch: Building with Python from First Principles.\" ISBN: 935213902X. [7] Arjovsky, M., et al. (2017). \"Wasserstein GAN.\" arXiv preprint arXiv:1701.07875.
Copyright
Copyright © 2025 Ravi Bhagat, Mrunal Bhasarkar, Bhadarge Akash, Bodke Sachin S, Nikita Pande. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET66826
Publish Date : 2025-02-04
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

